Scanning Electron Microscopy (SEM)
X-Ray Diffraction (XRD)
|
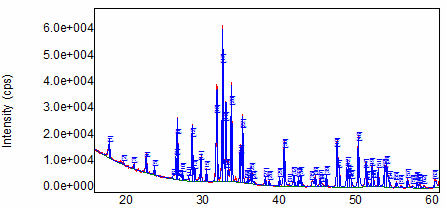
The XRD was used to determine the phase and purity of the hydroxyapatite produced from our synthesis. Each crystalline solid has a unique X-ray signature, which may be used as a "fingerprint" for its identification. Once the material has been identified, X-ray crystallography may be used to determine its structure. Analysis confirmed our synthesized hydroxyapatite to be 99% pure.
|
Dynamic Light Scattering (DLS)
|
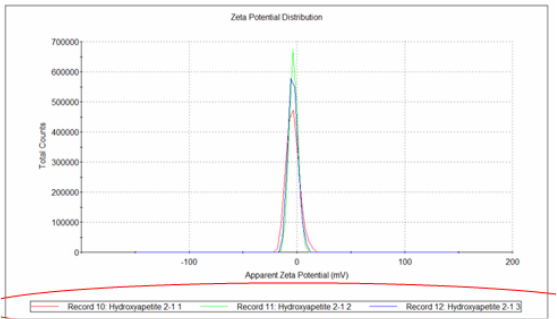
Following ball milling, our particles of hydroxyapatite were submitted to DLS testing for particle size characterization. A solution was prepared containing our sample dispersed in a non-reactive medium. Using polarized light and calculating the diffraction, we can find the average size of our particles. We did this for 3 different sets of samples, and the results are shown on the left.
|
Thermogravimetric Analysis (TGA)
|
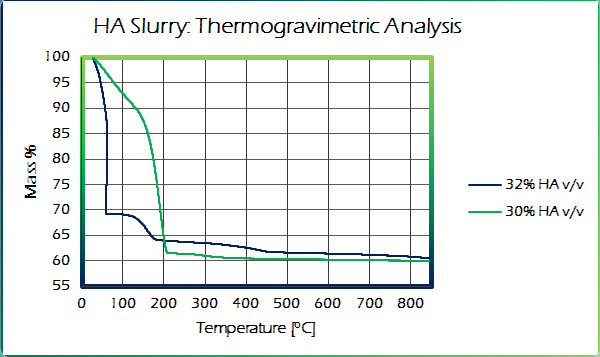
A TGA analysis was performed to better understand the thermal degradation of our slurry. Besides yielding information on drying rate and mass loss, this analysis also yielded the proper burnout temperature. The mass loss was then converted into volumetric loss and used to help calculate proper layer height during printing.
|
Cytotoxicity Analysis
|
Cytotoxicity is an extremely important test for implantable devices. This test analyses the capacity of cells grow into the surface of the sample. A positive result is necessary in order to bring our products to market. As seen in the image, our product is completely non-toxic, with the cells able to proliferate on the sample surface.
|
Aqueous Degradation
|
The human blood is a aqueous solution rich in ions and cells. This creates a harsh micro environment where things are constantly attacked and being degraded. Leaving our samples in a aqueous ion enriched solution, we attempted to produce an environment similar to what the implant will experience in the human body. Through future studies, we will obtain definitive results regarding the degradation rate of OsteoForge products.
|
Flexural Strength
|
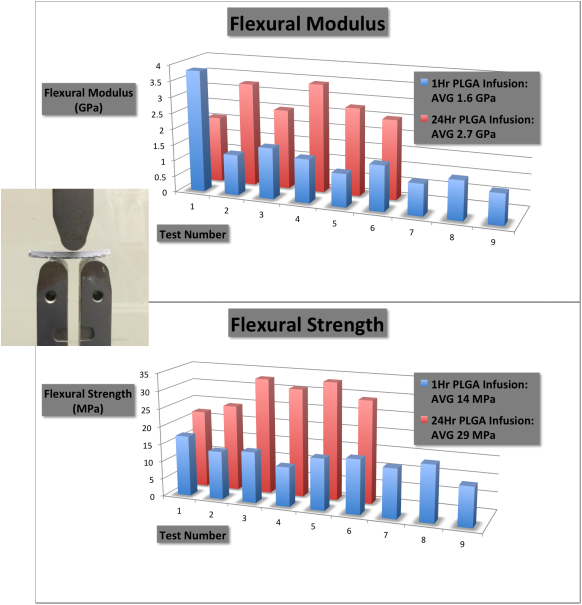
The 3-point bend simulates a realistic situation where the bone experiences tensile, compressive, and shear forces simultaneously. An example of our flexural test is shown on the far left.
Two sample sets were tested, each with a different polymer infusion time. The results show that the polymer infusion has an important role in slowing crack propagation through the ceramic. The flexural strength of bone ranges between 10 and 20 MPa with an elastic modulus of up to 0.5 - 30 GPa. Our product has an average flexural strength of 29 MPa with an elastic modulus of approximately 2.7 GPa. While slightly more robust than cancellous bone, OsteoForge products are somewhat less robust than cortical bone. But as our product will be used in both areas, an intermediate result is, in fact, desirable for mimicking bone's natural properties. |